

As a market leader in automated ultrasound reprocessing systems, Nanosonics’ trophon® technology helps protect patients by delivering consistent high level disinfection (HLD) of ultrasound probes with every automated cycle.
The trophon3 device is manufactured in Australia by Nanosonics and distributed exclusively by Biodecon in New Zealand.
‘Sonically activated’ H₂O₂ mist generates many free radicals with oxidative properties to kill bacteria, fungi, viruses, mycobacteria and spores
trophon technology is designed to provide HLD coverage of complex ultrasound probe morphologies in real world clinical settings.
Internal sensors monitor temperature, H₂O₂ mist volume, and H₂O₂ flow rate, while sophisticated software controls all aspects of the process to deliver effective automated HLD with every cycle
Operators are not exposed to chemicals, with gloves the only PPE required for use
No need for additional safety precautions (e.g., eyewash stations, ventilation cupboards) typically associated with some other reprocessing solutions
Low odour profile is suitable for continuous use in point of care settings
At completion of each HLD cycle, residual H₂O₂ is blown out of the disinfection chamber and passes through destructors, where it is broken down into environmentally friendly oxygen and water
Natural and rapid decomposition of H₂O₂ ensures no harmful residues persist on probes or in the environment
Nanosonics trophon technology meets or exceeds industry standards and guidelines for ultrasound probe HLD to combat healthcare-associated infections and protect patients globally.
This includes testing that meets the required EN, ASTM and AOAC standards to demonstrate:
Eliminates an extended range of clinically relevant pathogens including multidrug resistant bacteria, blood borne viruses, and sexually transmitted pathogens
There are no solutions on the market with broader efficacy than trophon
Automated trophon devices deliver consistent H₂O₂ disinfectant concentration and contact time to ensure antimicrobial efficacy, with an integrated monitoring system to confirm HLD is achieved with every cycle.
Comprehensive industry-leading program to approved probe compatibility with trophon technology
Every ultrasound probe listed as compatible with trophon devices has undergone rigorous testing to receive written approval and endorsement from the original equipment manufacturer (OEM).
Ultrasound probes are typically tested for 4,000 cycles in trophon devices
All probes listed as compatible are jointly approved by ultrasound probe manufacturers

You can find the full list of probes compatible with the trophon devices here.
To meet local accreditation requirements, traceability processes must be compliant with evidence-based standards and guidelines. The trophon®3 device uses AcuTrace radiofrequency identification (RFID) technology to electronically capture high-level disinfection (HLD) data across the reprocessing workflow in accordance with requirements in the USA, CAN, ANZ, UK, and EU.
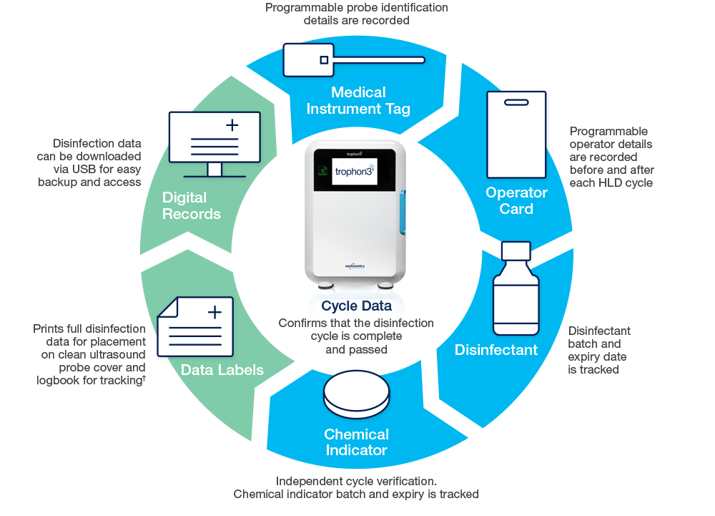
AcuTrace technology captures HLD data, which is accessible on your trophon3 device to support audit readiness via different user traceability methods:

NanoNebulant is a proprietary disinfectant liquid with 35% hydrogen peroxide chemistry to achieve effective high-level disinfection of the entire ultrasound probe including the shaft and handle. Incorporates AcuTrace technology to store batch and expiry details directly into trophon3.
The trophon Chemical Indicator responds to a combination of specified time, disinfectant dosage and disinfectant concentration. These critical parameters need to be met for the chemical indicator to change to a pass colour.
This qualitative colour change provides independent confirmation of success of each disinfection cycle. Indicator packaging incorporates AcuTrace technology to record and store batch and expiry details directly into trophon3.
Incorporates AcuTrace technology to digitally capture operator interaction details, stored directly on your trophon3.
Incorporates AcuTrace technology to digitally capture ultrasound probe details, stored directly on your trophon3.
AcuTrace PLUS makes the disinfection records stored on the trophon2 device accessible to other software applications over a network. Configure your IT systems* to centrally backup all trophon devices on the network.
* Custom API middleware required to enable this capability. All connectivity, configuration and integration with customer IT systems is the responsibility of the customer.
The trophon printer is a convenient traceability solution, printing high quality trophon disinfection labels each and every cycle.
High quality printer labels with a minimum 7-year life span, appropriate for medical records.
Simplifies and systematises record-keeping while providing evidence of quality HLD reprocessing at health care facilities.
A convenient option for installing trophon3 where there are space constraints.
Custom designed for secure, horizontal mounting of the trophon Printer to a wall. Includes a cable tidy area for convenience.
Makes trophon fully mobile for convenient point of care use and adaptability where there are space constraints.
Designed for securely attaching the trophon Printer to the trophon Cart, in an easily accessible, convenient location.
Reusable accessory designed specifically for the high-level disinfection of wireless ultrasound probes.
Footnotes
Abbreviations: AOAC, Association of Official Analytical Collaboration; ANZ, Australia and New Zealand; EU, European Union; ASTM, American Society for Testing and Materials; CE, European Commission; DICOM, Digital Imaging and Communications in Medicine; EN, European Standard; FDA, United States Food and Drug Administration; H₂O₂, hydrogen peroxide; HLD, high-level disinfection; PAC, Picture Archiving and Communication Systems; PPE, personal protective equipment; OEM, original equipment manufacturer; UPS, uninterruptable power supply.
References: 1. Vickery K, et al. J Infect Public Health. 2014;7(2):153–160. 2. Ryndock E, et al. J Med Virol. 2016;88(6):1076–1080. 3. Becker B, et al. GMS Hyg Infect Control. 2017;12:Doc02. 4. AORN 2018. High-Level Disinfection. In: AORN Guidelines for Perioperative Practice. Denver, CO. 5. ANSI/AAMI ST58:2013 Chemical sterilization and high-level disinfection in health care facilities. 6. CDC 2008 Guideline for Disinfection and Sterilization in Healthcare Facilities. 7. AS 5369:2023 Reprocessing of reusable medical devices and other devices in health and non-health related facilities. 8. Australasian Society for Ultrasound in Medicine (ASUM). Australas J Ultrasound Med. 2017;20(1):30–40. 9. Australian Commission on Safety and Quality in Health Care. National Safety and Quality Health Service Standards, second edition. November 2017. 10. CAN/CSA-Z314-18. Canadian Medical Device Reprocessing. February 2018. 11. Health Service Executive. Quality Improvement Division (2017). HSE Guidance for Decontamination of Semi‐critical Ultrasound Probes; Semi‐invasive and Non‐invasive Ultrasound Probes. 12. Health Protection Scotland (2016). NHS Scotland Guidance for Decontamination of Semi-Critical Ultrasound Probes; Semi-invasive and Non-invasive Ultrasound Probes. 13. Welsh Health Technical Memorandum (2014). WHTM 01-06 - Decontamination of flexible endoscopes Part C: Operational management. 14. Department of Health (UK) (2016). Health Technical Memorandum 01-06 Part C Operational management. 15. Ministère des affaires sociales et de la santé (2016). INSTRUCTION N° DGOS/PF2/DGS/VSS1/2016/220 du 4 juillet 2016 relative à relative au traitement des endoscopes souples thermosensibles à canaux au sein des lieux de soins. 16. Kommission für Krankenhaushygiene und Infektionsprävention and Bundesinstitut für Arzneimittel und Medizinprodukte. Anforderungen an die Hygiene bei der Aufbereitung von Medizinprodukten.